Jump To Topic
DePuy Lawsuit Information
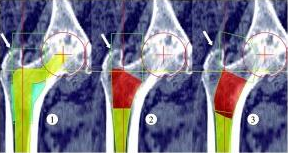
The Johnson & Johnson Depuy artificial hip implant has been recalled due to adverse event reports that between 12% and 13% of the Johnson And Johnson DePuy hip implants had to be surgically remow and replaced with new implants. Neither DePuy nor its parent company, Johnson & Johnson, is disclosing what is wrong with the implants. Some Orthopaedic surgeons have suggested that the problem may be the design of the cup that goes in the pelvis. The DePuy cup is shallower that cups in other models of hip implants and is harder for surgeons to implant. The recall is based on an unacceptably high percentage of hip implant failures requiring an additional surgery called a revision surgery.
Problems experienced by patients with Johnson & Johnson DePuy hip implants a.k.a ASR XL hip implants.
Update on DePuy ASR Hip Replacement Settlement
- – DePuyASR Total Hip Replacement Settlement Update – June 1st, 2014
- – ASR Hip Implant Settlement Update – May 15th, 2014
- – DePuyASR Hip Implant Litigation Update – May 3rd, 2014
The problems that are being reported by people with the DePuy ASR XL hip implants include:
- Loose cups in the hip socket causing pain and instability,
- Allergic reactions to metallic debris in the hip joint,
- Development of soft tissue masses called pseudo tumors that form as a reaction to exposure to toxins from the metal debris within the hip.
- No physical symptoms but blood tests that show high levels of microscopic metal particles debris that might cause problems later.
How are Johnson & Johnson Depuy ASR XL hip implants different than other Hip implants on the Market?
The DePuy ASR XL hip implants are metal on metal implants. Sometimes these are referred to in the industry as MoM implants. That means that the inside of the cup is metal and the ball on the end of the femur is metal. The binding of metal on metal where the cup is not properly aligned may be creating the metal debris that has been found in the hip joint and causing some of the problems to patients. Other hip implants on the market are ceramic liners on the cups and ceramic balls on the femur or polyethylene liners in the cups with ceramic balls on the femurs. Surgeons make decisions on the type of implant based upon many different factors. Some of these factors are age and activity level of the patient. Surgeons also consider the size of the patient and the condition of the patients existing bone in recommending types of hip implants.
How long have the DePuy ASR XL implants been on the market in the U.S.?
The DePuy ASR hips were first implanted in the U.S. in 2006. The FDA cleared them for sale in the U.S. in 2005 but did not perform a detailed review called a premarket approval or PMA. The FDA relied on DePuy’s representation that the implants were similar enough to other products already on the market that an in depth premarket approval review was unnecessary.
Why could DePuy be liable to pay compensation in a hip implant lawsuit to people who received DePuy ASR implants and have continuing pain, disability, or require another surgery?
While the laws vary in the 50 United States, every state provides for compensation for pain, suffering, disability, loss of income, and medical expenses to some degree. All states laws can impose liability upon a manufacturer of a product such as a hip implant if the manufacturer was negligent in the design or manufacture of a product that causes injury. Many states have strict product liability laws that can impose liability in a lawsuit if the risks of the product design doesn’t justify any additional benefit of the product design. Further, DePuy can be held liable for compensation in a lawsuit if it is shown that they knew of the failures of the product and failed to promptly recall the product to protect patients.
If Johnson & Johnson DePuy offers to pay my medical expenses for another surgery can I still bring a claim for compensation or file a lawsuit?
Yes, manufacturers sometimes offer to pay medical expenses for an additional surgery where they have sold a defective product. An offer by the Company does not mean that you have to accept it. Patients are free to bring a claim or lawsuit for all of the damages sustained not just medical expenses. This choice is a personal decision. However, you should get a legal opinion from an attorney independent from the Company before making a decision on this.
Sometimes manufacturers contact patients with defective medical implants through the doctors and suggest that settling with the Company directly and not paying a lawyer will save them money. However, most lawyers, including myself, will give free opinions on your case before you decide.
Additionally, medical expenses for an additional surgery would be usually covered by insurance or Medicare for those fortunate enough to have insurance.
Does Bringing a Lawsuit against DePuy involve suing my Doctor?
In most cases, product liability lawsuits for defective hip implants do not involve suing doctors. You should get legal advice and make this decision with a lawyer’s help. Manufacturers of bad hip implants in the past have sometimes suggested that it was the surgeons fault to deflect blame from themselves but this is usually for publicity and most of the time the doctors are not parties to the case. The doctors are the customers of DePuy and DePuy has a close relationship with most of the doctors who implant their products so it is very unlikely that DePuy would want to doctor in the case.

