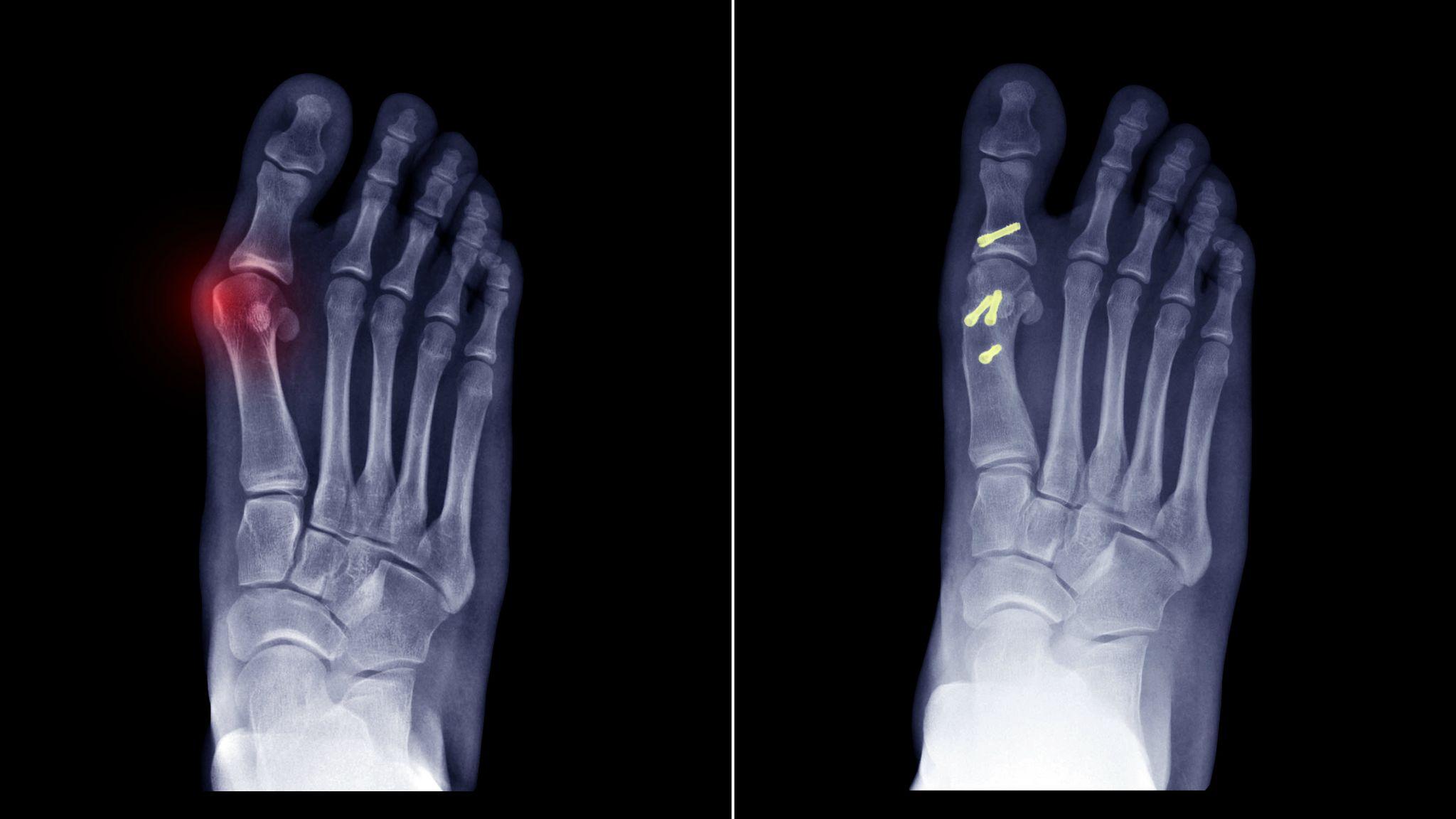
Can I Bring a Lawsuit for Compensation for My Cartiva Toe Implant?
The Cartiva synthetic cartilage implant was the subject of a Class 2 FDA recall in October 2024 due to an unusually high failure rate. Many
Read More
Catholic Church Bankruptcy Cases: The Diocese of Alexandria, Louisiana
The Diocese of Alexandria, Louisiana, filed for Chapter 11 bankruptcy on October 31, 2025. This does not mean the Diocese has no money to compensate
Read More
How Statutes of Limitation Affect Justice for Survivors of Clergy Abuse
For many survivors of clergy sexual abuse, finding the strength to speak out can take decades. Unfortunately, the legal system often operates on strict timelines
Read More
The Genesis and Development of the Exactech Orthopedic Hip and Knee Implant Recall Lawsuits
The first Exactech hip implant failure case came into my office before any of the FDA product recalls. I was very familiar with hip implants
Read More
The Louisiana Supreme Court Reverses Itself and Allows Justice for Survivors of Childhood Sex Abuse.
On June 12th, 2024, the Louisiana Supreme Court released an opinion upholding the statute of limitations look back window that the Louisiana legislature passed in
Read More
Pacific Islands New Area Where Priests Are Sent After They Abuse Children
In the wake of Pope Francis’ trip to Papua New Guinea, the New York Times is reporting on the number of priests accused of sexual
Read More
List of Priests Accused of Abuse in Baltimore
In recent years, the U.S. Catholic Church has been shaken by numerous accusations of child sexual abuse, with some cases relating to incidents occurring as
Read More
The Recent Maryland Attorney General Report on Clergy Abuse: What You Need to Know
The State of Maryland has long been committed to ensuring the safety and well-being of its citizens. In a recent push towards transparency and justice,
Read More
Understanding the Exactech Recall
Exactech, a noted manufacturer of joint replacement devices, is currently involved in a wide-ranging recall relating to a large number of its implants that were
Read More
Paraquat Lawsuit Update 2023
Paraquat is a widely used pesticide that has been shown to be highly toxic to human beings. Ingesting just a small amount of this chemical
Read More
