 The mortality rate for untreated pulmonary emboli is 30%. Sudden death is the first symptom in about one-quarter (25%) of people who have a PE. Pulmonary emboli affect approximately 900,000 people each year in the United States. Of those 900,000, 10-30% will die within a month of experiencing the embolism.
The mortality rate for untreated pulmonary emboli is 30%. Sudden death is the first symptom in about one-quarter (25%) of people who have a PE. Pulmonary emboli affect approximately 900,000 people each year in the United States. Of those 900,000, 10-30% will die within a month of experiencing the embolism.
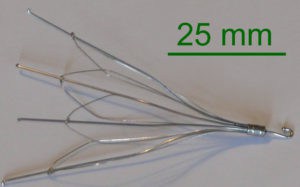
The standard treatment is anticoagulation, but in situations where anti-coagulation is not an option, interruption of the inferior vena cava is considered. IVC filters are small, cage-like devices that are inserted into the inferior vena cava to capture blood clots and prevent them from reaching the lungs. The inferior vena cava is the main vessel returning blood from the lower half of the body to the heart. IVC filters are designed to be permanent implants although some of these devices may have the option to be removed.
These filters, both the permanent and the retrievable, have been shown to have their own set of dangerous, sometimes deadly side effects. The FDA has received reports of adverse events and product problems associated with IVC filters. Types of reports include device migration, filter fracture, embolization (movement of the entire filter or fracture fragments to the heart or lungs), perforation of the IVC, and difficulty removing the device. Some of these events led to adverse clinical outcomes. These types of events may be related to how long the filter has been implanted. Other known long-term risks associated with IVC filters include lower limb deep vein thrombosis and IVC occlusion. For patients with retrievable filters, some complications may be avoided if the filter can be removed once the risk of pulmonary embolism has subsided. The FDA is concerned that retrievable IVC filters, when placed for a short-term risk of pulmonary embolism, are not always removed once the risk subsides.
Retrievable filters are also difficult to remove without causing harm to the patient. In a recent study of 240 patients who underwent placement of IVC filters, only 73 (30.4%) had documented plans for filter removal and of 62 who underwent attempted filter retrieval, 25.8% of filters could not be successfully removed. The recommended duration of use varies for each manufacturer, but the longer a filter is in place, the more difficult it is to remove. Filters that are not removed stay in place and may be associated with potential complications.
These filter risks led the FDA to issue a warning in 2010. The FDA stated that IVC filters may fail, leading to parts of the filters to migrate through a patient’s body – leading to serious injury or even death. The FDA advised doctors to remove IVC filters from patients when the risk of pulmonary embolism subsides.
As I wrote earlier, the retrieval process can be just as dangerous as the risk of filter fracture or migration. The IVC filters are not safe products and have caused severe, life-threatening injuries to consumers. If you or a loved one has been injured by an IVC filter, call me for a free consultation.

